World
New catalytic strategies boost alkene synthesis

Two new catalytic methods offer ways to tackle longstanding challenges in alkene synthesis. Alkenes are essential chemical building blocks used in the manufacture of everyday items such as plastics and rubber, as well as more complex molecules like pharmaceutical ingredients. One of the new methods improves the efficiency of making long-chain alkenes via the Fischer–Tropsch process, while the other provides a general way to make complex alkenes from carboxylic acid, alcohol or alkane starting materials.
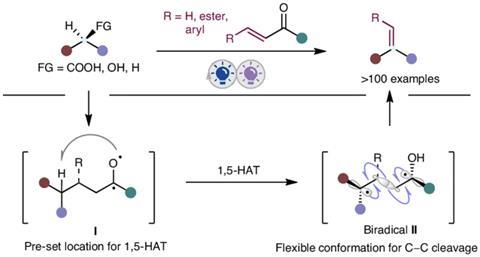
Alkenes are crucial intermediates in organic synthesis, materials science, and the pharmaceutical industry. However, their production often requires harsh conditions and specific starting materials. ‘To overcome these limitations, we have introduced a photocatalytic conjugate addition step that not only enhances selectivity but also expands the substrate scope to include carboxylic acids, alcohols, and alkanes for efficient alkene synthesis,’ explains Hao Zeng, a researcher at the National University of Singapore (NUS).
The new photocatalytic strategy harnesses visible light to drive the transformations under mild conditions.1 It involves a two-step, one-pot process that features a radical addition followed by fragmentation. ‘We employ vinyl ketones as radical acceptors to generate a saturated carbonyl intermediate,’ says Zeng. The resulting carbonyl intermediate undergoes selective Norrish type photoreaction, resulting in the desired alkene products.
‘The methodology allows for the synthesis of a wide variety of alkenes, including α-alkenes and internal alkenes, by coupling with substituted vinyl aryl ketones,’ says Zeng. He points out that the process can be applied to complex molecules containing multiple functional groups, and could be especially useful for late-stage diversification strategies used by drug discovery chemists. Ming Joo Koh, an organic chemist at NUS who wasn’t involved in the project, notes that the reaction complements existing methods for preparing alkenes. ‘There is good innovation involved here,’ he says. However, Koh points out that one limitation is that the method ‘isn’t suitable for making stereochemically defined alkenes’.
Meanwhile, chemists at Eindhoven University of Technology have boosted the Fischer–Tropsch conversion of synthesis gas (syngas) – a mixture of carbon monoxide and hydrogen – into alkenes by using ‘phase-pure’ iron carbide catalysts.2 This phase purity refers to the way the researchers have maximised the active form of the catalyst, while minimising the generation of iron oxides that drive unwanted side-reactions. The resulting system enables high carbon monoxide conversion under mild conditions and prevents the production of carbon dioxide waste.

‘Iron catalysts suffer from carbon dioxide side-product formation, often constituting almost half of the product mixture in terms of carbon atoms – meaning a significant loss of valuable carbon,’ says Eindhoven’s Emiel Hensen. Hensen’s team reports that its new catalytic system achieves activities ‘1–2 orders higher’ than dedicated Fischer–Tropsch catalysts achieve at even higher temperatures.
‘The much higher activity presented here means that it may start competing with more active, yet also much more expensive, cobalt catalysts,’ explains Hensen. He adds that systems like this may become even more important in future decarbonisation efforts by enabling syngas derived from waste plastic and biomass to be converted into useful materials.
Gopinthan Sankar, a solid-state chemist at University College London in the UK, notes that the work addresses the production of short-chain terminal alkenes. ‘This promising work may be further investigated to make this [Fischer Tropsch reaction] process more efficient,’ he says. However, Koh points out that ‘it remains to be seen if this current work can be realistically commercialised on an industrial scale’.
‘Both [projects] are addressing the production of olefins which are good platform chemicals for further processing to obtain a range of products,’ observes Sankar. Both Zeng and Hensen’s groups are now focused on developing their respective methods. They hope to eventually scale up the processes for use in industry, where they could be used in a range of sectors, including fine chemical production and synthesis of pharmaceuticals and agrochemicals.










